| Line 7: | Line 7: | ||
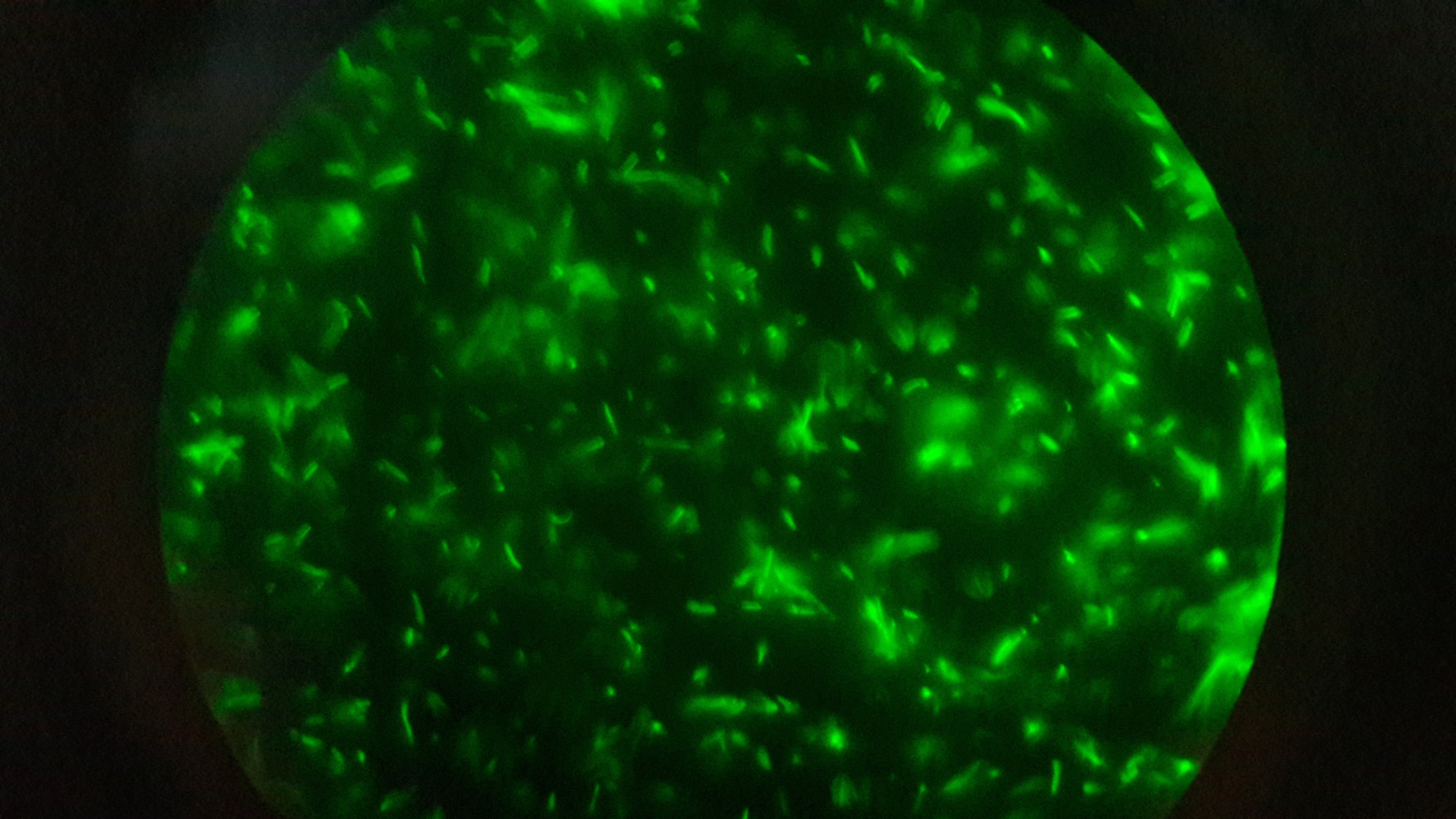
| − | [[File:T--Aix-Marseille--fluorondfort.jpeg|thumb|]] | + | [[File:T--Aix-Marseille--fluorondfort.jpeg|thumb|Fluorescence of GFP of E.Coli]] |
Revision as of 16:57, 11 October 2017
InterLab
How close can the numbers be when fluorescence is measured all around the world ?
In order to support the interlab approach, our team participate to the Fourth International InterLab Measurement Study. We are glad to contribute to this studie based on reliable and repeatable measurement. The challenge this year consist in etablishing a GFP measurement protocol that can be used produce comparable GFP measurements on plate readers. Furthermore, teams are also going to test some RBS devices that are intended to make gene expression more precise and reliable.
Materials and Methods:
Used plasmids
Devices from Interlab Measurement Kit
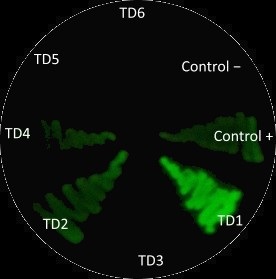
- Positive control
- Negative control
- Test Device 1:J23101.BCD2.E0040.B0015
- Test Device 2:J23106.BCD2.E0040.B0015
- Test Device 3:J23117.BCD2.E0040.B0015
- Test Device 4:J23101+I13504
- Test Device 5:J23106+I13504
- Test Device 6:J23117+I13504
Used strain
E. coli E5alpha
Used material
1ml LUDOX H20 96 well plate,black with flat bottom fluorescein 10ml 1xPBS LB (Luria Bertani) media Chloramphenicol (stock concentration 25mg/mL dissolved in EtOh - working stock 25 ug/ml) 50 ml Falcon tube Incubator at 37°C 1.5 ml eppendorf tube Ice bucket Pipettes
Used machines
- Tecan's Infinite®
- Confocal microscope
Used software
Link protocoles
Description :
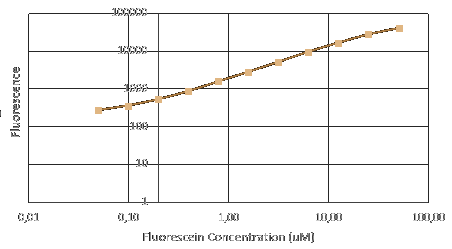
We used plasmids containing different promoter from the 2017 distribution kit and transfected them into E.coli. Before measuring fluorescence expression, we used LUDOX and fluorescein to calibrate the TECAN. We prepared a dilution series of fluorescein in 4 duplicates and measure the fluorescence in a 96 well plate in a plate reader. By measuring these in all standard modes, we generated a standard curve of fluorescence for fluorescein concentration. We used this to correct cell based readings to an equivalent fluorescein concentration and convert this into a concentration of GFP.
Results
| Hour 0: | Neg. Control | Pos. Control | Device 1 | Device 2 | Device 3 | Device 4 | Device 5 | Device 6 | LB + Chlor (blank) |
| Colony 1, Replicate 1 | 156 | 165 | 167 | 177 | 157 | 159 | 153 | 153 | 147 |
| Colony 1, Replicate 2 | 155 | 161 | 166 | 161 | 151 | 160 | 156 | 158 | 154 |
| Colony 1, Replicate 3 | 158 | 157 | 164 | 152 | 144 | 159 | 151 | 155 | 147 |
| Colony 1, Replicate 4 | 157 | 161 | 165 | 155 | 150 | 156 | 155 | 155 | 155 |
| Colony 2, Replicate 1 | 158 | 159 | 163 | 178 | 154 | 158 | 158 | 153 | 152 |
| Colony 2, Replicate 2 | 89 | 104 | 97 | 112 | 93 | 99 | 100 | 90 | 91 |
| Colony 2, Replicate 3 | 94 | 103 | 99 | 109 | 93 | 103 | 99 | 97 | 89 |
| Colony 2, Replicate 4 | 92 | 102 | 101 | 80 | 89 | 95 | 94 | 97 | 90 |
| Hour 2: | Neg. Control | Pos. Control | Device 1 | Device 2 | Device 3 | Device 4 | Device 5 | Device 6 | LB + Chlor (blank) |
| Colony 1, Replicate 1 | 102 | 132 | 124 | 121 | 98 | 117 | 112 | 99 | 93 |
| Colony 1, Replicate 2 | 101 | 126 | 120 | 125 | 99 | 109 | 118 | 96 | 93 |
| Colony 1, Replicate 3 | 98 | 126 | 121 | 116 | 98 | 113 | 115 | 91 | 84 |
| Colony 1, Replicate 4 | 99 | 128 | 122 | 118 | 91 | 111 | 115 | 93 | 92 |
| Colony 2, Replicate 1 | 105 | 133 | 118 | 139 | 101 | 111 | 116 | 102 | 93 |
| Colony 2, Replicate 2 | 99 | 128 | 113 | 139 | 103 | 110 | 114 | 104 | 95 |
| Colony 2, Replicate 3 | 100 | 130 | 110 | 131 | 102 | 109 | 113 | 102 | 97 |
| Colony 2, Replicate 4 | 96 | 132 | 116 | 133 | 101 | 116 | 112 | 99 | 97 |
| Hour 4: | Neg. Control | Pos. Control | Device 1 | Device 2 | Device 3 | Device 4 | Device 5 | Device 6 | LB + Chlor (blank) |
| Colony 1, Replicate 1 | 109 | 228 | 168 | 250 | 105 | 147 | 152 | 105 | 93 |
| Colony 1, Replicate 2 | 112 | 236 | 173 | 254 | 101 | 146 | 166 | 106 | 92 |
| Colony 1, Replicate 3 | 112 | 243 | 174 | 259 | 101 | 154 | 162 | 108 | 85 |
| Colony 1, Replicate 4 | 114 | 242 | 171 | 255 | 101 | 153 | 149 | 108 | 92 |
| Colony 2, Replicate 1 | 103 | 224 | 157 | 262 | 99 | 151 | 156 | 106 | 102 |
| Colony 2, Replicate 2 | 109 | 239 | 165 | 274 | 104 | 157 | 161 | 108 | 93 |
| Colony 2, Replicate 3 | 110 | 239 | 161 | 263 | 105 | 162 | 159 | 109 | 91 |
| Colony 2, Replicate 4 | 108 | 241 | 167 | 272 | 106 | 166 | 169 | 112 | 95 |
| Hour 6: | Neg. Control | Pos. Control | Device 1 | Device 2 | Device 3 | Device 4 | Device 5 | Device 6 | LB + Chlor (blank) |
| Colony 1, Replicate 1 | 148 | 362 | 348 | 481 | 147 | 343 | 211 | 142 | 120 |
| Colony 1, Replicate 2 | 156 | 378 | 351 | 510 | 137 | 347 | 219 | 148 | 120 |
| Colony 1, Replicate 3 | 150 | 382 | 318 | 497 | 138 | 355 | 218 | 153 | 120 |
| Colony 1, Replicate 4 | 154 | 385 | 354 | 479 | 138 | 358 | 210 | 151 | 130 |
| Colony 2, Replicate 1 | 149 | 354 | 336 | 493 | 136 | 375 | 210 | 150 | 130 |
| Colony 2, Replicate 2 | 149 | 391 | 365 | 564 | 146 | 399 | 193 | 147 | 128 |
| Colony 2, Replicate 3 | 154 | 367 | 355 | 507 | 140 | 416 | 210 | 149 | 130 |
| Colony 2, Replicate 4 | 153 | 385 | 359 | 560 | 148 | 417 | 232 | 151 | 138 |