| Line 33: | Line 33: | ||
The Gaussian chip (Fig.4) was designed to test if our exogenous genes will influence their olfactory receptor neuron pair (preference and repulsion to some chemical odors). | The Gaussian chip (Fig.4) was designed to test if our exogenous genes will influence their olfactory receptor neuron pair (preference and repulsion to some chemical odors). | ||
| − | {{SUSTech_Image_Center_8 | filename= | + | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Microfuildics--gstd.png|width=600px|caption=<B>Fig.4 The Gaussian Chip</B>}} |
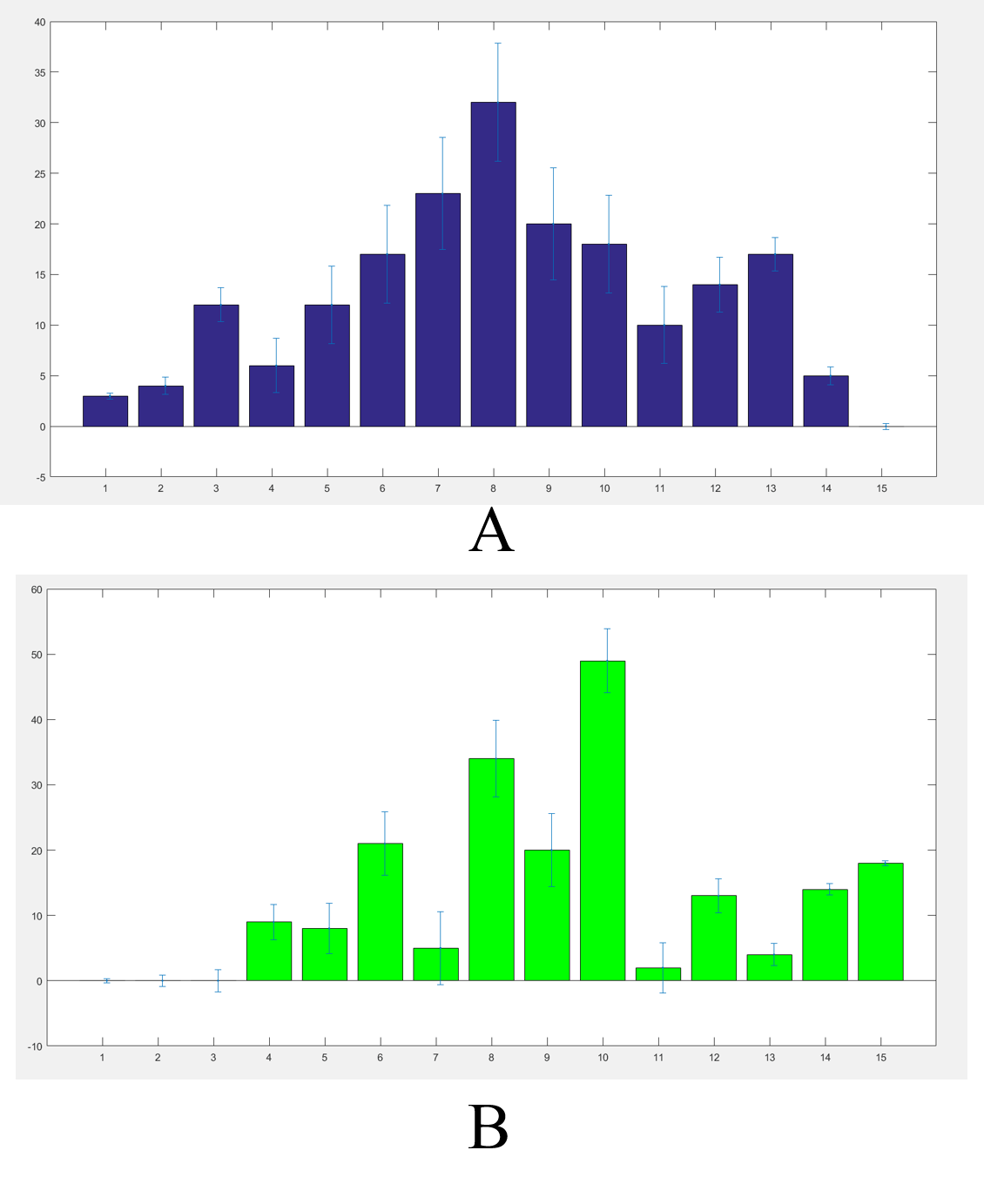
We got the worms’ distributions(Fig.5) after several experiments for the wildtype worms and our experimental worms with or without the chemicals (Fig.6). | We got the worms’ distributions(Fig.5) after several experiments for the wildtype worms and our experimental worms with or without the chemicals (Fig.6). | ||
| − | {{SUSTech_Image_Center_8 | filename= | + | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Microfuildics--gsdata.png|width=600px|caption=<B>Fig.5 The distribution of the worms. A) The distribution of the worms without diacetyl. B) The distribution of the worms with diacetyl in channel A.</B>}} |
| − | {{SUSTech_Image_Center_8 | filename= | + | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Microfuildics--gsgif.gif|width=600px|caption=<B>Fig.6 The Experiment Process of the Gaussian Chip</B>}} |
The immobilization chip was deigned to immobilize the C. elegans in worm traps or parallel channels for worm imaging and ethological experiments. | The immobilization chip was deigned to immobilize the C. elegans in worm traps or parallel channels for worm imaging and ethological experiments. | ||
| − | {{SUSTech_Image_Center_8 | filename= | + | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Microfuildics--guding.png|width=600px|caption=<B>Fig.7 The Immobilization Chip</B>}} |
We could immobilize the worms in the worm traps (Fig.8) and watch the neuronal activity (Fig.9) successfully using fluorescence microscope (Nikon eclipse Ti). In addition, we could stimulate the Odr10::CoChR::GEM-GECO::mCherry worms to be active from the low state (Video.1). Unfortunately, we cannot see the clear neuro images in str1::Chrimson::GEM-GECO::GFP worms using fluorescence microscope but we can use confocal microscope to observe the neuro successfully. | We could immobilize the worms in the worm traps (Fig.8) and watch the neuronal activity (Fig.9) successfully using fluorescence microscope (Nikon eclipse Ti). In addition, we could stimulate the Odr10::CoChR::GEM-GECO::mCherry worms to be active from the low state (Video.1). Unfortunately, we cannot see the clear neuro images in str1::Chrimson::GEM-GECO::GFP worms using fluorescence microscope but we can use confocal microscope to observe the neuro successfully. | ||
| − | {{SUSTech_Image_Center_8 | filename= | + | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Microfuildics--guding1.jpeg|width=600px|caption=<B>Fig.8 The Worms in Immobilization Chip.</B>}} |
| − | {{SUSTech_Image_Center_8 | filename= | + | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Microfuildics--mcherry.png|width=600px|caption=<B>Fig.9 The mCherry of the AWA Neuro in the Odr10::CoChR::GEM-GECO::mCherry Worm..</B>}} |
= References = | = References = | ||
Revision as of 23:08, 24 October 2017
Results
Project
Optical Experiments Resutls
Microfluidic Experiments Resutls
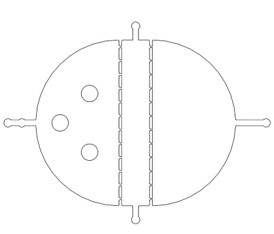
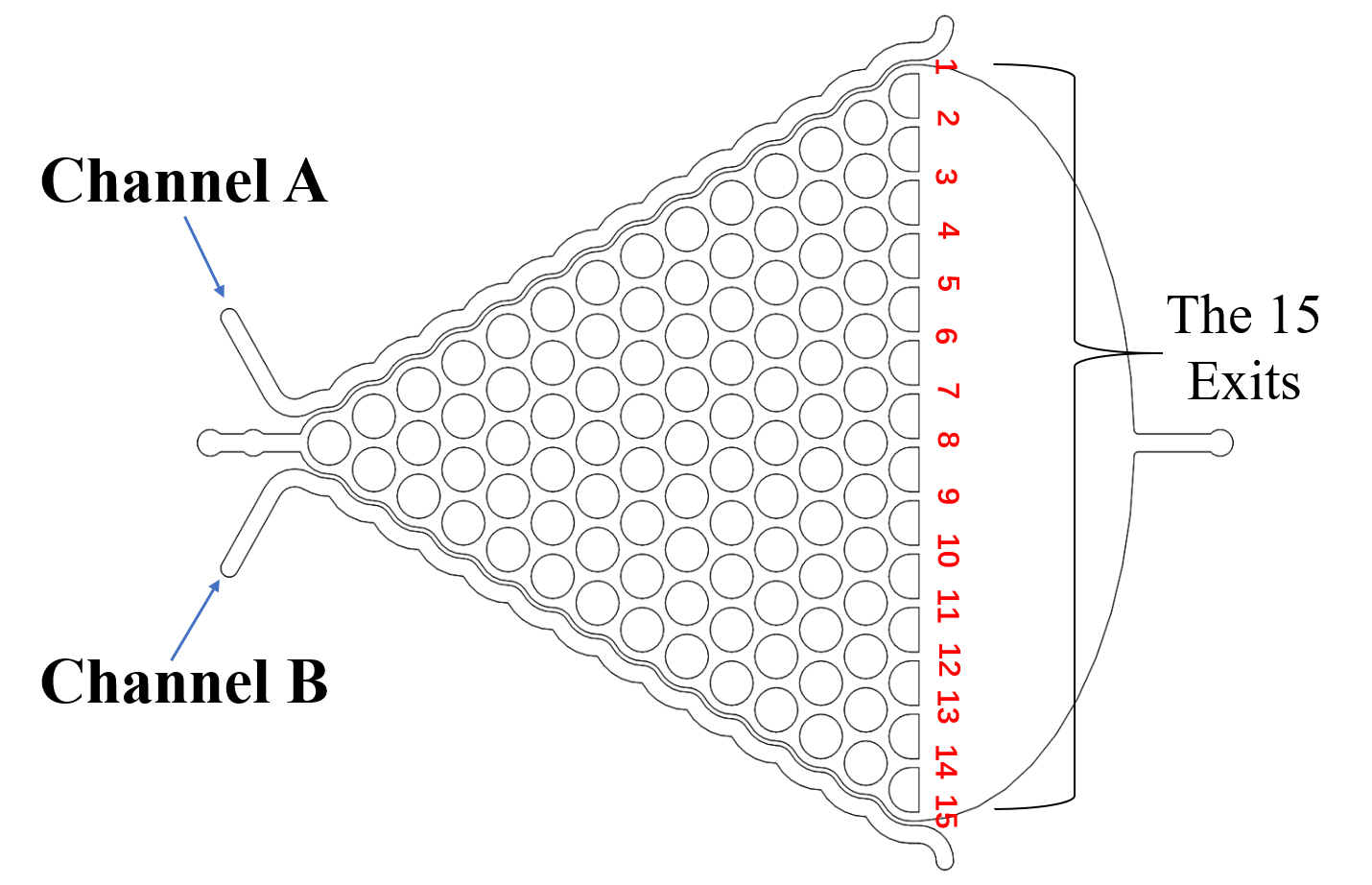
Microfluidics were divide into three parts: the selective chip; the Gaussian chip; and the immobilization chip. The selective chip was designed to select the C. elegans with the appropriate size (Fig.1).
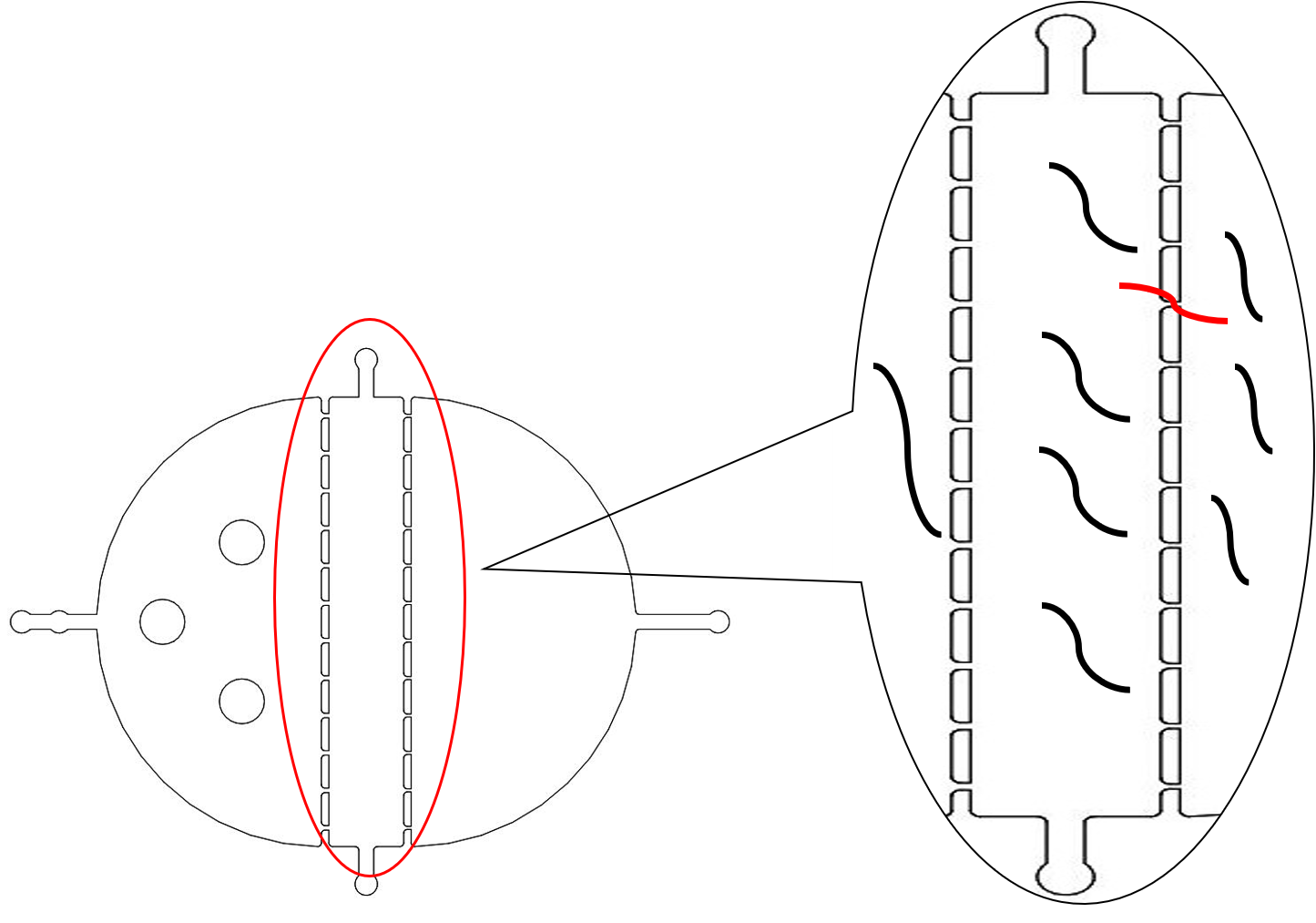
We found that the chip only had 12 fences (Fig.2), but our next experiments needed a large number of the C. elegans which were at the same stage, so the efficiency of the selective chip was very low. In addition, the C. elegans have flexible body, some of the suitable size worms would still go through the second fences (Fig.2).
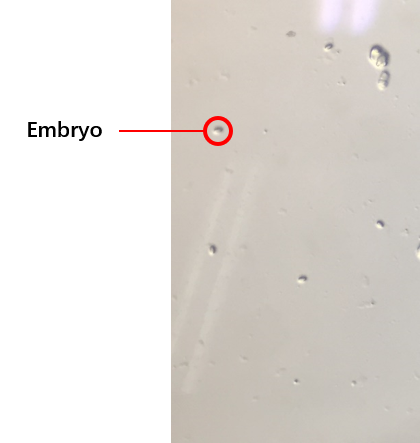
Then, the C. elegans’ synchronization was utilized to get a large number of worms at the same stage. We got the embryos (Fig.3) from the old worms so that the worms would be at the same stage because of the hatches of the embryos were at the same time. We selected several conditions of the synchronization, finally, we could get the worms at the same stage. The synchronous rate ((The number of the wroms at L4)/(All the worms)×100%) could reach to about 80%.
The Gaussian chip (Fig.4) was designed to test if our exogenous genes will influence their olfactory receptor neuron pair (preference and repulsion to some chemical odors).
We got the worms’ distributions(Fig.5) after several experiments for the wildtype worms and our experimental worms with or without the chemicals (Fig.6).
The immobilization chip was deigned to immobilize the C. elegans in worm traps or parallel channels for worm imaging and ethological experiments.
We could immobilize the worms in the worm traps (Fig.8) and watch the neuronal activity (Fig.9) successfully using fluorescence microscope (Nikon eclipse Ti). In addition, we could stimulate the Odr10::CoChR::GEM-GECO::mCherry worms to be active from the low state (Video.1). Unfortunately, we cannot see the clear neuro images in str1::Chrimson::GEM-GECO::GFP worms using fluorescence microscope but we can use confocal microscope to observe the neuro successfully.
References