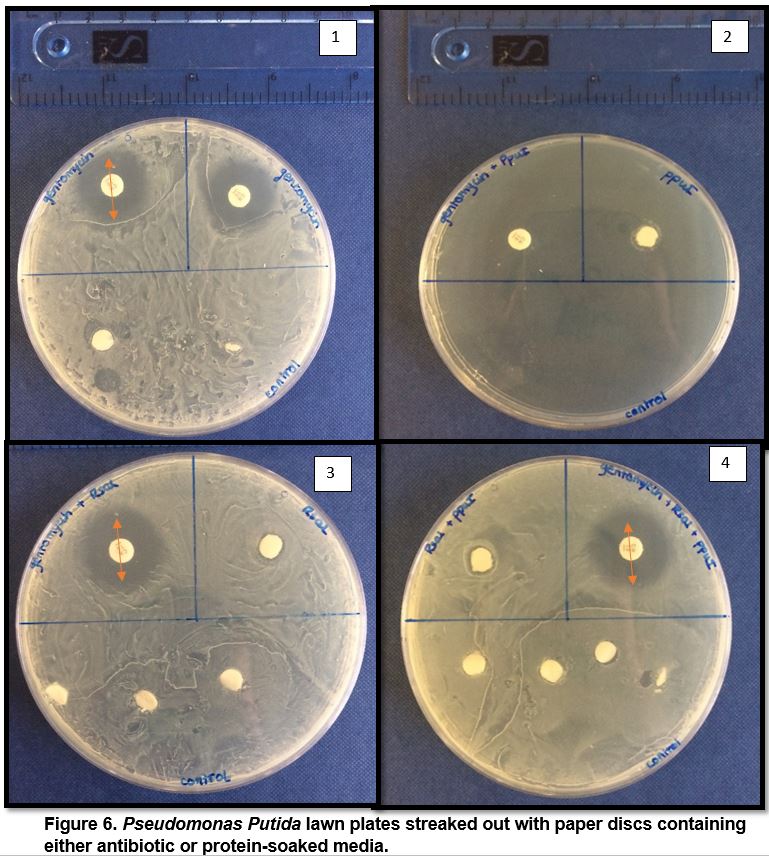
|
|
| Line 4: |
Line 4: |
| | <div class="Vwrapper"> | | <div class="Vwrapper"> |
| | <h1>Proof of concept</h1> | | <h1>Proof of concept</h1> |
| − |
| |
| − | <p>
| |
| − | To evaluate the implication of our transformed genes in biofilms, we
| |
| − | cultured <em>P.putida </em>biofilms in the presence of our genes to see
| |
| − | whether they would induce a positive or negative impact on biofilm
| |
| − | formation.
| |
| − | </p>
| |
| − | <p>
| |
| − | To do so, 8 plates were made: one with AHL coded by PpuI, one with
| |
| − | synthetic AHL diluted in DMSO, one with synthetic AHL diluted in water, one
| |
| − | with AHL receptor-inhibitor coded by Rsal 2, one with AHL
| |
| − | receptor-inhibitor coded by Rsal 4, one with periplasmic protease coded by
| |
| − | LapG, and one with DMSO, and one with PSB1C3 as a negative control.
| |
| − | </p>
| |
| − | <p>
| |
| − | To extract the product of these genes, we inoculated universal bottles with
| |
| − | colonies from each transformation and left them to incubate overnight in a
| |
| − | shaking incubator at 37 C.
| |
| − | </p>
| |
| − | <p>
| |
| − | At the same time, <em>P putida</em> was inoculated in 5mL of LB broth and
| |
| − | left to incubate overnight in the same shaking incubator at 37 C.
| |
| − | </p>
| |
| − | <p>
| |
| − | The following day, <em>P.putida</em> was diluted 1:100 in LB broth then 100
| |
| − | mL of this solution were pipetted in columns 3-12, and 200 mL were pipetted
| |
| − | into column 2 . In all wells of column 1, 200 mL of LB broth were added. In
| |
| − | total, 8 plates were set up.
| |
| − | </p>
| |
| − | <p>
| |
| − | The universal bottles inoculated with transformed <em>E.coli</em> were
| |
| − | centrifuged at 1,500 rpm for 20 min. The supernatant, or LB broth, was
| |
| − | conserved and the pellet, containing the bacterial cells, was discarded. 10
| |
| − | eppendorf tubes were set up, labelled 1-10. In tube 1, 1,000 mL of the
| |
| − | supernatant was added. In tubes 2-10, 900 mL of LB broth were added. We
| |
| − | then proceeded to perform a serial dilution, by pipetting 100 mL from tube
| |
| − | 1 to tube 2, then 100 mL from tube 2 to tube 3, and so on up to tube 10.
| |
| − | </p>
| |
| − | <p>
| |
| − | This serial dilution was performed for the products of PpuI, Rsal 2, Rsal
| |
| − | 4, LapG.
| |
| − | </p>
| |
| − | <p>
| |
| − | We also serial diluted synthetic AHL, first into DMSO and then into water.
| |
| − | Once again, we set up 8 eppendorf tubes. Tubes 2-8 were loaded with 900 mL
| |
| − | of DMSO. Tube 1 contained 1000 mL of a concentration of 10mM of AHL. We
| |
| − | serial diluted from tube 1 to 8, transferring 100 mL from one tube to the
| |
| − | next.
| |
| − | </p>
| |
| − | <p>
| |
| − | As for the water dilution, tube 1 remained the same as above (10 mM of
| |
| − | AHL). Distilled water was pipetted into tubes 2 to 8, 900 mL into each. 100
| |
| − | mL were transferred from tube 1 to tube 2, and so on.
| |
| − | </p>
| |
| − | <p>
| |
| − | As a control, we serial diluted DMSO, with tube 1 containing 100% DMSO, and
| |
| − | the remaining 9 tubes containing 900 mL of water and 100 mL from the
| |
| − | previous tube.
| |
| − | </p>
| |
| − | <p>
| |
| − | To column 3 in each of the plates, we added 100 mL from Eppendorf tube 10
| |
| − | (a different one for each plate). Unless we were adding synthetic AHL, in
| |
| − | which case we started with Eppendorf tube 8. We worked our way down the
| |
| − | series of eppendorfs – the concentration was lowest in column 3 and highest
| |
| − | in column 12 (or column 10 in the case of synthetic AHL).
| |
| − | </p>
| |
| − | <p>
| |
| − | The plates were left to incubate at 37 C overnight. The OD600 was read
| |
| − | before staining the plates with 0.1% crystal violet. Once stained, the
| |
| − | plates were read at 590 nm.
| |
| − | </p>
| |
| | | | |
| | <img src="https://static.igem.org/mediawiki/2017/7/7b/Antibiotic_1.jpeg" width"100%" height="100%"> </br> | | <img src="https://static.igem.org/mediawiki/2017/7/7b/Antibiotic_1.jpeg" width"100%" height="100%"> </br> |