| Line 12: | Line 12: | ||
== Optical Experiments Resutls == | == Optical Experiments Resutls == | ||
| − | {{SUSTech_Image_Center_fill-width | filename=T--SUSTech_Shenzhen--3dworm.gif|width=800px|caption= | + | {{SUSTech_Image_Center_fill-width | filename=T--SUSTech_Shenzhen--3dworm.gif|width=800px|caption=Fig. 1 The 3D reconstruction of the ord-10::CoChR::GEM-GECO::mCherry worms' mCherry in AWA neuron. The most}} |
== Microfluidic Experiments == | == Microfluidic Experiments == | ||
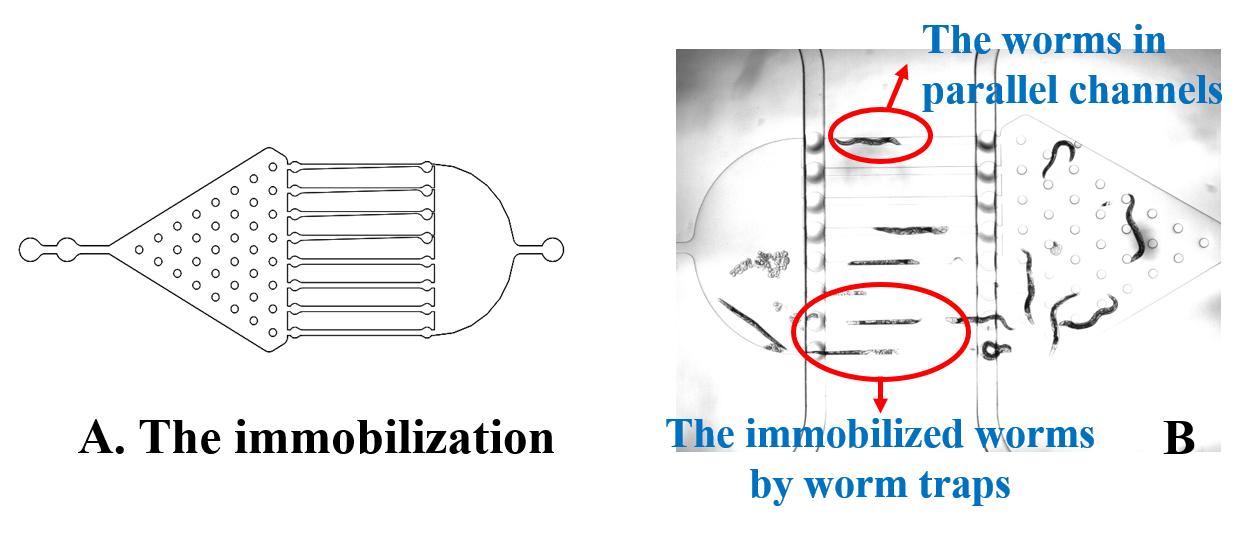
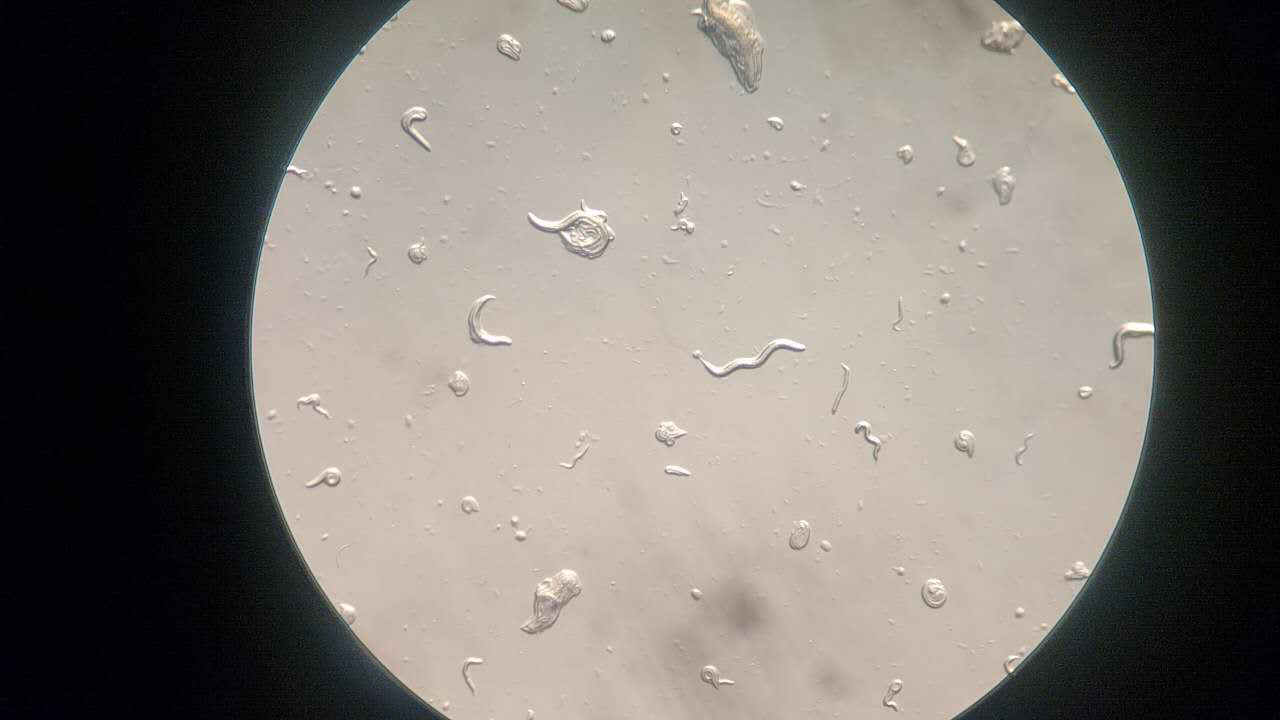
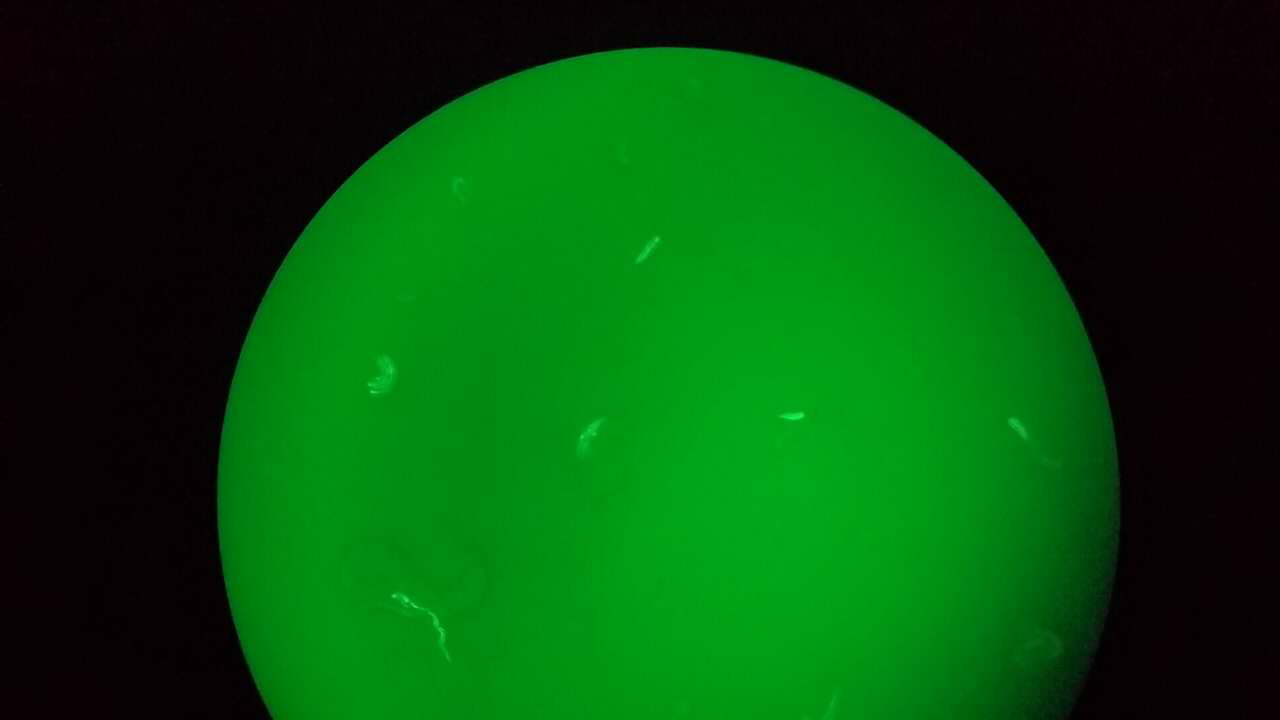
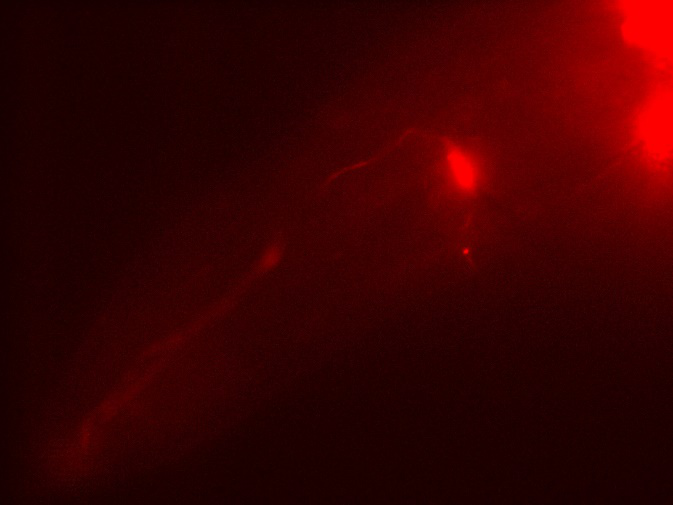
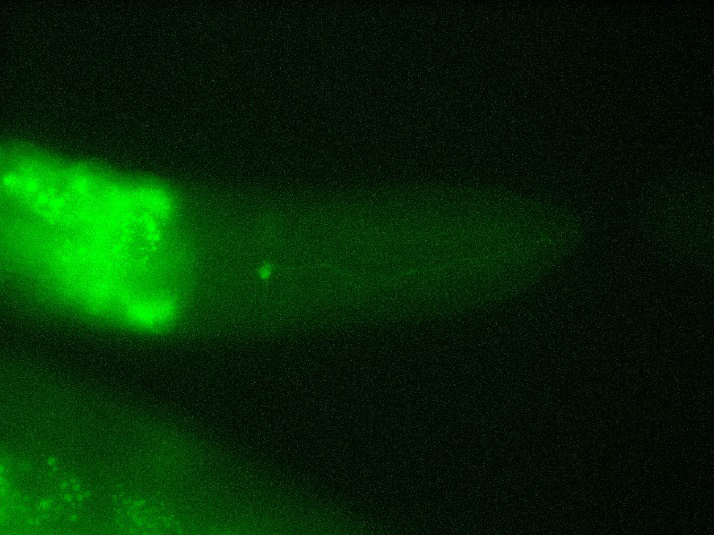
Here, we fixed the <i>Caenorhabditis elegans</i> in <i>the Immobilization Chip</i> to observe the Odr10::CoChR::GEM-GECO::mCherry worms under the fluorescence microscope and saw the neuronal activity successfully, which can confirm that the worms can express our target genes. We also put the Odr10::CoChR::GEM-GECO::mCherry worms into the chip, after a few minutes the worms would be inactive, then we can "wake up" the worms by the blue light. | Here, we fixed the <i>Caenorhabditis elegans</i> in <i>the Immobilization Chip</i> to observe the Odr10::CoChR::GEM-GECO::mCherry worms under the fluorescence microscope and saw the neuronal activity successfully, which can confirm that the worms can express our target genes. We also put the Odr10::CoChR::GEM-GECO::mCherry worms into the chip, after a few minutes the worms would be inactive, then we can "wake up" the worms by the blue light. | ||
| − | + | ||
| − | {{SUSTech_Image_Center_fill-width | filename=T--SUSTech_Shenzhen--Microfuildics--result00.png|width=800px|caption=<B>Fig.1 A. <i>The Immobilization Chip</i>. B. The worms in <i>the Immobilization Chip</i>.</B>}} | + | {{SUSTech_Image_Center_fill-width | filename=T--SUSTech_Shenzhen--Microfuildics--result00.png|width=800px|caption=<B>Fig.1 A. <i>The Immobilization Chip</i>. B. The worms in <i>the Immobilization Chip</i>.</B>}} |
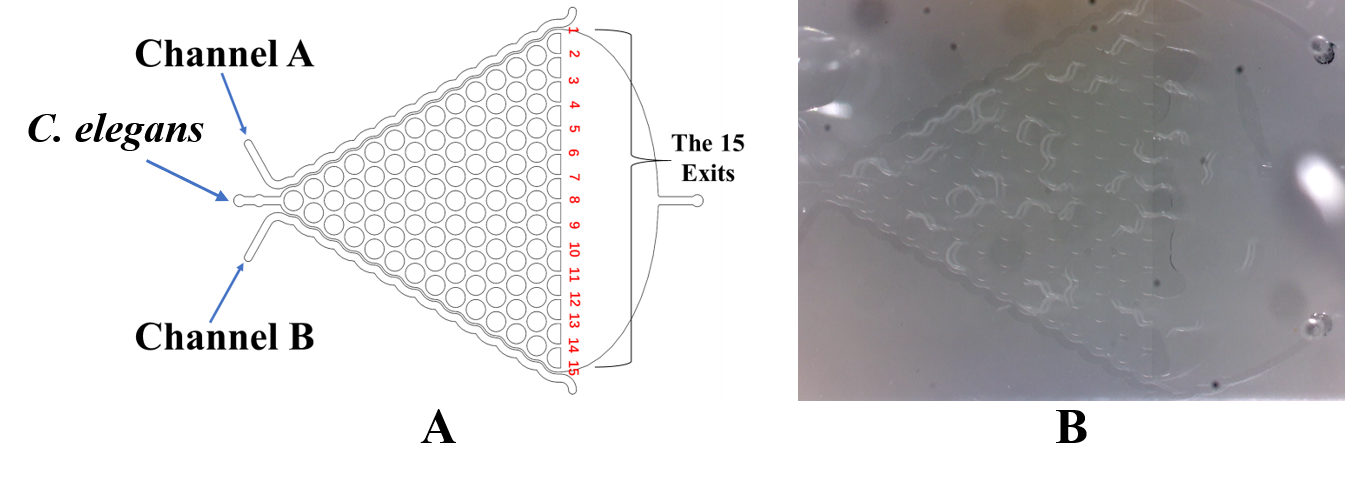
Then, we demonstrated that the insertion did not damage the olfactory receptor neuron pairs of the worms by testing their response to diacetyl and 2-nonanone in <i>the Gaussian Plate</i>. | Then, we demonstrated that the insertion did not damage the olfactory receptor neuron pairs of the worms by testing their response to diacetyl and 2-nonanone in <i>the Gaussian Plate</i>. | ||
| Line 26: | Line 26: | ||
<html><a target="_blank" href="https://2017.igem.org/Team:SUSTech_Shenzhen/Results/Microfluidic" class="btn btn-default"><i class="ion-arrow-right-c"></i> See Details</a></html> | <html><a target="_blank" href="https://2017.igem.org/Team:SUSTech_Shenzhen/Results/Microfluidic" class="btn btn-default"><i class="ion-arrow-right-c"></i> See Details</a></html> | ||
| − | == Behavioral Experiments == | + | == Behavioral Experiments == |
Here, we confirmed that the Odr10::CoChR::GEM-GECO::mCherry worms could sense the blue light by inducing the Odr10::CoChR::GEM-GECO::mCherry worms to crawl a cycle on NGM plate. The Odr10::CoChR::GEM-GECO::mCherry worms could follow the blue light spot just like the attract of the food. | Here, we confirmed that the Odr10::CoChR::GEM-GECO::mCherry worms could sense the blue light by inducing the Odr10::CoChR::GEM-GECO::mCherry worms to crawl a cycle on NGM plate. The Odr10::CoChR::GEM-GECO::mCherry worms could follow the blue light spot just like the attract of the food. | ||
| Line 34: | Line 34: | ||
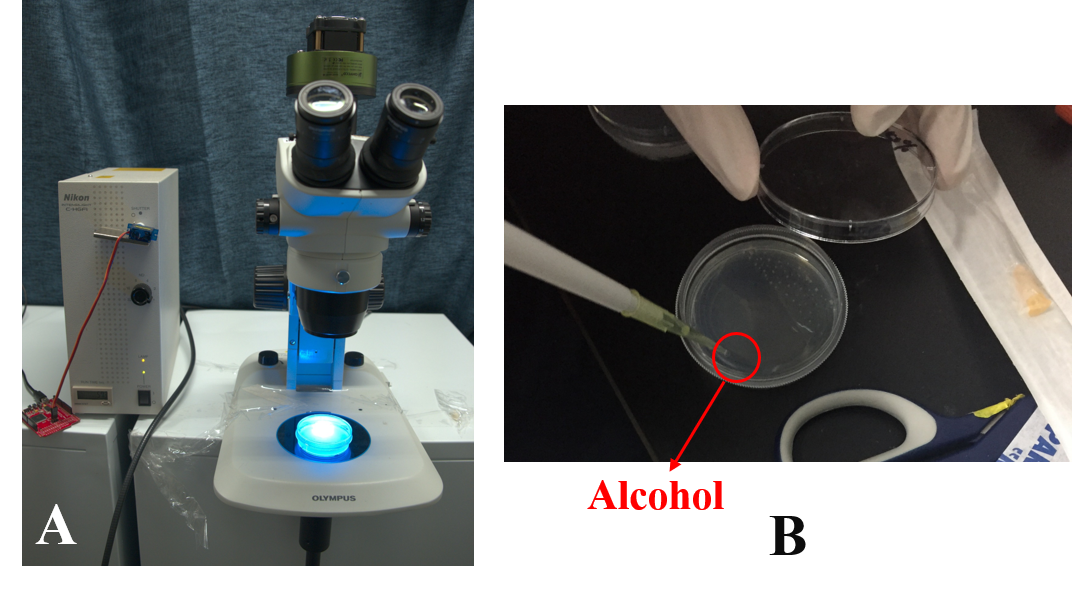
Then, in order to study the worms' learning ability we put the worms in alcohol layer on the NGM plate and stimulated them by the blue light at the same time. After 2 hours' training we found that the worms could crawl towards to the alcohol. | Then, in order to study the worms' learning ability we put the worms in alcohol layer on the NGM plate and stimulated them by the blue light at the same time. After 2 hours' training we found that the worms could crawl towards to the alcohol. | ||
| − | {{SUSTech_Image_Center_fill-width | filename=T--SUSTech_Shenzhen--Microfuildics--result013.png|width=800px|caption=<B>Fig.4 A.The device made by mercury lamp and microscope for training the worms.(More details in [https://2017.igem.org/Team:SUSTech_Shenzhen/Hardware# | + | {{SUSTech_Image_Center_fill-width | filename=T--SUSTech_Shenzhen--Microfuildics--result013.png|width=800px|caption=<B>Fig.4 A.The device made by mercury lamp and microscope for training the worms.(More details in [https://2017.igem.org/Team:SUSTech_Shenzhen/Hardware#Light_Modulator Hardware] B. Using the alcohol to induce the worms.</B>}} |
<html><a target="_blank" href="https://2017.igem.org/Team:SUSTech_Shenzhen/Results/Behavior" class="btn btn-default"><i class="ion-arrow-right-c"></i> See Details</a></html> | <html><a target="_blank" href="https://2017.igem.org/Team:SUSTech_Shenzhen/Results/Behavior" class="btn btn-default"><i class="ion-arrow-right-c"></i> See Details</a></html> | ||
| Line 53: | Line 53: | ||
In order to get <i>C. elegans</i> strains with the preference to blue lights and the aversion to red lights, we used miniMos injection to inject our plasmids in to worms for expression. | In order to get <i>C. elegans</i> strains with the preference to blue lights and the aversion to red lights, we used miniMos injection to inject our plasmids in to worms for expression. | ||
| − | On July.7, we microinjected 20 worms with ord-10:: | + | On July.7, we microinjected 20 worms with ord-10::CoChR::GEM-GECO::mCherry and 20 worms with Str-1::Chrimson::GEM-GECO::GFP. After 3 days, for each kinds of worms, we obtained more than 6 free-moving F1 with fluorescences. Then, 10 days after microinjection, we did heat shock to screen stable inheritance worms. For worms with ord-10::CoChR::GEM-GECO::mCherry, one plate successfully survived more than 30 worms without GFP(a selective marker), but none of worms with Str-1::Chrimson::GEM-GECO::GFP survived. In addition, we did mapping experiments and demonstrated that ord-10::CoChR::GEM-GECO::mCherry had successfully inserted in chromosome 1. |
{{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Results-MI1.png|width=800px|caption=<B>Fig.7 heat shock</B>}} | {{SUSTech_Image_Center_8 | filename=T--SUSTech_Shenzhen--Results-MI1.png|width=800px|caption=<B>Fig.7 heat shock</B>}} | ||
Revision as of 01:26, 2 November 2017
Results
Project
Contents
Optical Experiments Resutls
Microfluidic Experiments
Here, we fixed the Caenorhabditis elegans in the Immobilization Chip to observe the Odr10::CoChR::GEM-GECO::mCherry worms under the fluorescence microscope and saw the neuronal activity successfully, which can confirm that the worms can express our target genes. We also put the Odr10::CoChR::GEM-GECO::mCherry worms into the chip, after a few minutes the worms would be inactive, then we can "wake up" the worms by the blue light.
Then, we demonstrated that the insertion did not damage the olfactory receptor neuron pairs of the worms by testing their response to diacetyl and 2-nonanone in the Gaussian Plate.
Behavioral Experiments
Here, we confirmed that the Odr10::CoChR::GEM-GECO::mCherry worms could sense the blue light by inducing the Odr10::CoChR::GEM-GECO::mCherry worms to crawl a cycle on NGM plate. The Odr10::CoChR::GEM-GECO::mCherry worms could follow the blue light spot just like the attract of the food.
Then, in order to study the worms' learning ability we put the worms in alcohol layer on the NGM plate and stimulated them by the blue light at the same time. After 2 hours' training we found that the worms could crawl towards to the alcohol.
Plasmid Construction Results
According to the design of plasmid construction, we constructed ord-10::CoCHR::GEM-geco::mCherry and str-1::Chrimson::GEM-GECO::GFP fusion genes in backbone pCFJ909 successfully. The fusion gene segments were all be sequenced.
We also amplify B series plasmid in miniMos system for microinjection. We integrated ord-10::CoChR::GEM-GECO::mCherry and str-1::Chrimson::GEM-GECO::GFP fusion genes into C. elegans(Caenorhabditis elegans) by microinjection respectively.
Microinjection Results
In order to get C. elegans strains with the preference to blue lights and the aversion to red lights, we used miniMos injection to inject our plasmids in to worms for expression.
On July.7, we microinjected 20 worms with ord-10::CoChR::GEM-GECO::mCherry and 20 worms with Str-1::Chrimson::GEM-GECO::GFP. After 3 days, for each kinds of worms, we obtained more than 6 free-moving F1 with fluorescences. Then, 10 days after microinjection, we did heat shock to screen stable inheritance worms. For worms with ord-10::CoChR::GEM-GECO::mCherry, one plate successfully survived more than 30 worms without GFP(a selective marker), but none of worms with Str-1::Chrimson::GEM-GECO::GFP survived. In addition, we did mapping experiments and demonstrated that ord-10::CoChR::GEM-GECO::mCherry had successfully inserted in chromosome 1.
On August 1st, we microinject worms using Str-1::Chrimson::GEM-GECO::GFP. This time we injected 20 worms and also observed F1 phenotype after 3 days, picking up free-moving worms with RFP and did heat shock 9 days later. After heat shock, on August 21th, we got about 10 worms expressed plasmids without arrays, meaning that we obtained stable inheritance worms expressing Str-1::Chrimson::GEM-GECO::GFP.