Djcamenares (Talk | contribs) |
Djcamenares (Talk | contribs) (→Sequencing Verification) |
||
| Line 28: | Line 28: | ||
Text here about the PCR reactions to create the fragments for Gibson Assembly | Text here about the PCR reactions to create the fragments for Gibson Assembly | ||
| − | + | [[File:T--Kingsborough_NY--sequencing_comparison_K2268004_cI.png|600px|left]] | |
| − | > VF2 Primer result, reverse complement | + | [[File:T--Kingsborough_NY--sequencing_comparison_K2268004_lacZa.png|600px|left]] |
| + | |||
| + | <br clear=all> | ||
| + | |||
| + | Full sequencing results for construct K2268004 | ||
| + | |||
| + | <b> VF2 Primer result, reverse complement </b> | ||
| + | |||
| + | <font family="Courier New"> | ||
GGGNACGTTACGTTGCGCATTTAACTATTAGATGCGTCGAATCACGAGGCAGGAATTTCAGATAAAAAAAATCCTTAGCT | GGGNACGTTACGTTGCGCATTTAACTATTAGATGCGTCGAATCACGAGGCAGGAATTTCAGATAAAAAAAATCCTTAGCT | ||
TTCGCTAAGGATGATTTCTGGAATTCGCGGCCGCTTCTAGAGTAACACCGTGCGTGTTGACTATTTTACCTCTGGCGGTG | TTCGCTAAGGATGATTTCTGGAATTCGCGGCCGCTTCTAGAGTAACACCGTGCGTGTTGACTATTTTACCTCTGGCGGTG | ||
| Line 36: | Line 44: | ||
TGGGAAAACCCTGGCGTTACCCAACTTAATCGCCTTGCAGCACATCCCCCTTTCGCCAGCTGGCGTAATAGCGAAGAGGC | TGGGAAAACCCTGGCGTTACCCAACTTAATCGCCTTGCAGCACATCCCCCTTTCGCCAGCTGGCGTAATAGCGAAGAGGC | ||
CCGCACCGATCGCCCTTCCCAACAGTTGCGCAGCCTGAATGGCGAAT | CCGCACCGATCGCCCTTCCCAACAGTTGCGCAGCCTGAATGGCGAAT | ||
| + | </font> | ||
| + | <b> 3.2_LinkR Primer result, reverse complement </b> | ||
| − | > | + | <font family="Courier New"> |
ATCACCAGCTCCCGTCTTTCATTGCATACGAAATTCCGGAGAGCATTCATCAGGCGGGCA | ATCACCAGCTCCCGTCTTTCATTGCATACGAAATTCCGGAGAGCATTCATCAGGCGGGCA | ||
AGAAATGTGAATAAAGGCCGAATAAAACTTGTGCTTATTTTTCTTTACGGTCTTTAAAAA | AGAAATGTGAATAAAGGCCGAATAAAACTTGTGCTTATTTTTCTTTACGGTCTTTAAAAA | ||
| Line 53: | Line 63: | ||
ACAGAGGGCAGCTTGAATGGCGAATGGGCTTTGCCTGGTTTTTCGAATTCTTCCGCAACT | ACAGAGGGCAGCTTGAATGGCGAATGGGCTTTGCCTGGTTTTTCGAATTCTTCCGCAACT | ||
TACTGCGTAGCGNA | TACTGCGTAGCGNA | ||
| + | </font> | ||
| − | > 3.2_LinkF Primer result | + | <b> 3.2_LinkF Primer result </b> |
| + | |||
| + | <font family="Courier New"> | ||
NTCCATGATAAGTACTCGCACTTTCTCAGCAGTCATCAGTTCGCCGCGCCAAACGTCTCTTCAGGCCACTGACTAGCGAT | NTCCATGATAAGTACTCGCACTTTCTCAGCAGTCATCAGTTCGCCGCGCCAAACGTCTCTTCAGGCCACTGACTAGCGAT | ||
AACTTTCCCCACAACGGAACAACTCTCATTGCATGGGATCATTGGGTACTGTGGGTTTAGTGGTTGTAAAAACACCTGAC | AACTTTCCCCACAACGGAACAACTCTCATTGCATGGGATCATTGGGTACTGTGGGTTTAGTGGTTGTAAAAACACCTGAC | ||
| Line 70: | Line 83: | ||
GCAGGACTCGTAAAAGCCGCGTGCTGGCGATTCACAAGGCTCGCCCCCTGACGGCACTACAAAATCGACGCTCTAGGCAG | GCAGGACTCGTAAAAGCCGCGTGCTGGCGATTCACAAGGCTCGCCCCCTGACGGCACTACAAAATCGACGCTCTAGGCAG | ||
AGGTGCGAACCGCACGGACATAAAGGATCCAGC | AGGTGCGAACCGCACGGACATAAAGGATCCAGC | ||
| + | </font> | ||
| − | > VR Primer result, reverse complement | + | <b>* VR Primer result, reverse complement</b> |
| + | |||
| + | <font family="Courier New"> | ||
ATGATTCGGATCCCTGGCCGTGTTTACACGTTCGGAACTGGGAAACCCTGCGTACCAACT | ATGATTCGGATCCCTGGCCGTGTTTACACGTTCGGAACTGGGAAACCCTGCGTACCAACT | ||
AATCCCTTGCAGCACATCCCTTTCGCAGCTGCGTAATAGCGAAGAGGCCGCCCGATCGCC | AATCCCTTGCAGCACATCCCTTTCGCAGCTGCGTAATAGCGAAGAGGCCGCCCGATCGCC | ||
| Line 93: | Line 109: | ||
GCGTATTGGGCGCTCTGCCCCTCACTCGCTCCCTCTGGAAACTGCGGCAGTACGTTGCGC | GCGTATTGGGCGCTCTGCCCCTCACTCGCTCCCTCTGGAAACTGCGGCAGTACGTTGCGC | ||
GGAATGTAAGG | GGAATGTAAGG | ||
| + | </font> | ||
| + | |||
| + | The sequencing primers that were used included the 3.2 Link F Primer (ggtgccggaaagctggctggagtaataaTtattacgccaccagcgcatag) and 3.2 Link R Primer (ctatgcgctggtggcgtaataAttattactccagccagctttccggcacc) | ||
<br clear=all> | <br clear=all> | ||
Revision as of 02:59, 1 November 2017
Replace this with text describing the construction of our BioBricks (3.1 and 3.2) - in other words, the colony PCR and sequencing Data
PCR to Generate Fragments for Gibson Assembly
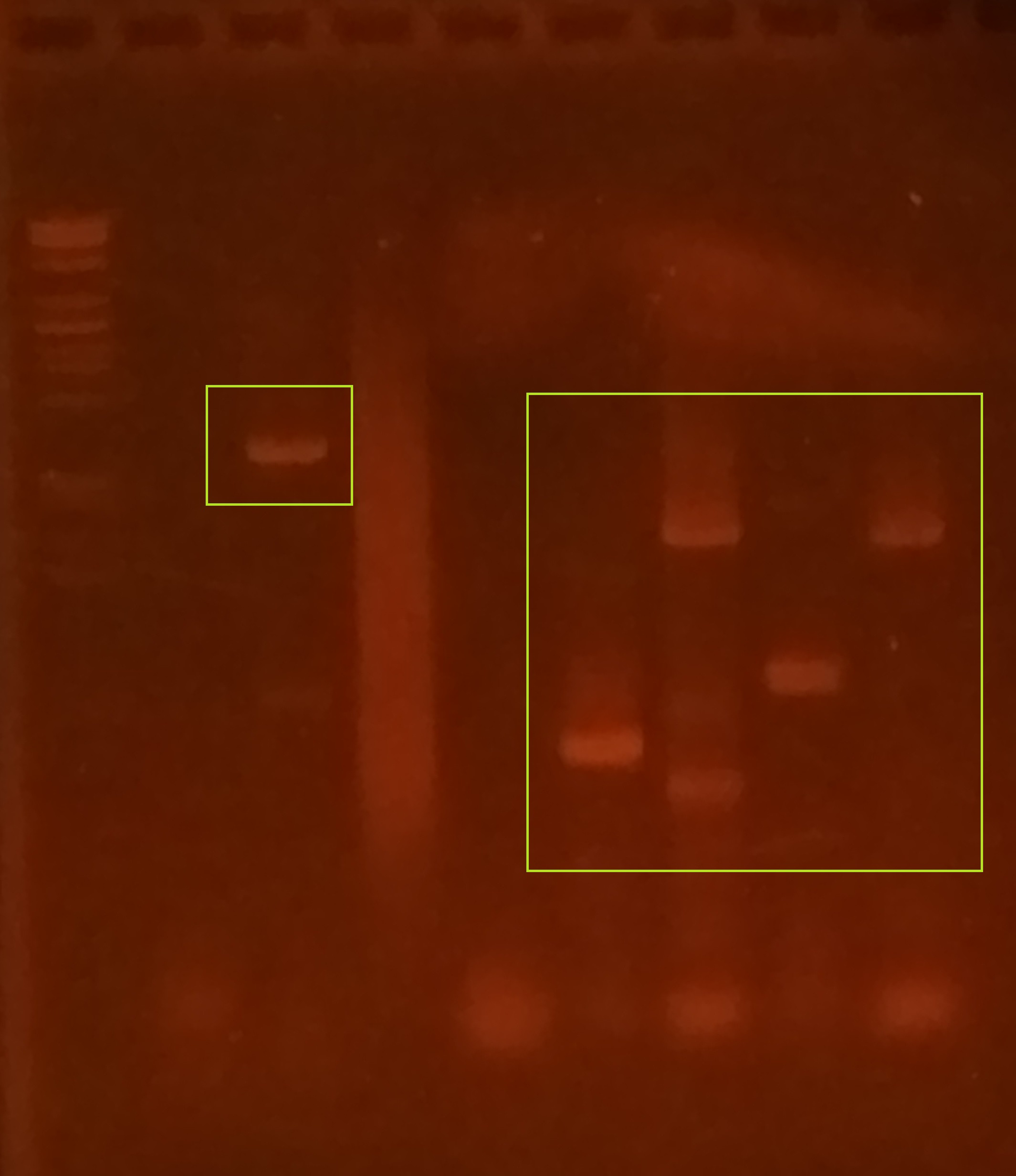
Fragments obtained from IDT to construct our plasmid designs were not suitable for Gibson Assembly. Therefore, we ordered primers and performed PCR to generate fragments with homology to each other and/or the pSB1C3 plasmid backbone. These fragments were resolved on an agarose gel (shown on the right).
The first lane contains our molecular weight marker: Lambda DNA digested with EcoRI and PstI. the first small yellow box shows amplification of a single fragment containing all the genes for construct K2268006 (MazF and cI repressor). The second yellow box shows the various fragments (representing LacZ-alpha, cI repressor, or MazF-ssrA with appropriate homologies) that were subsequently used in Gibson Assembly (after purification, of course!).
Colony PCR to Verify Clone is Correct
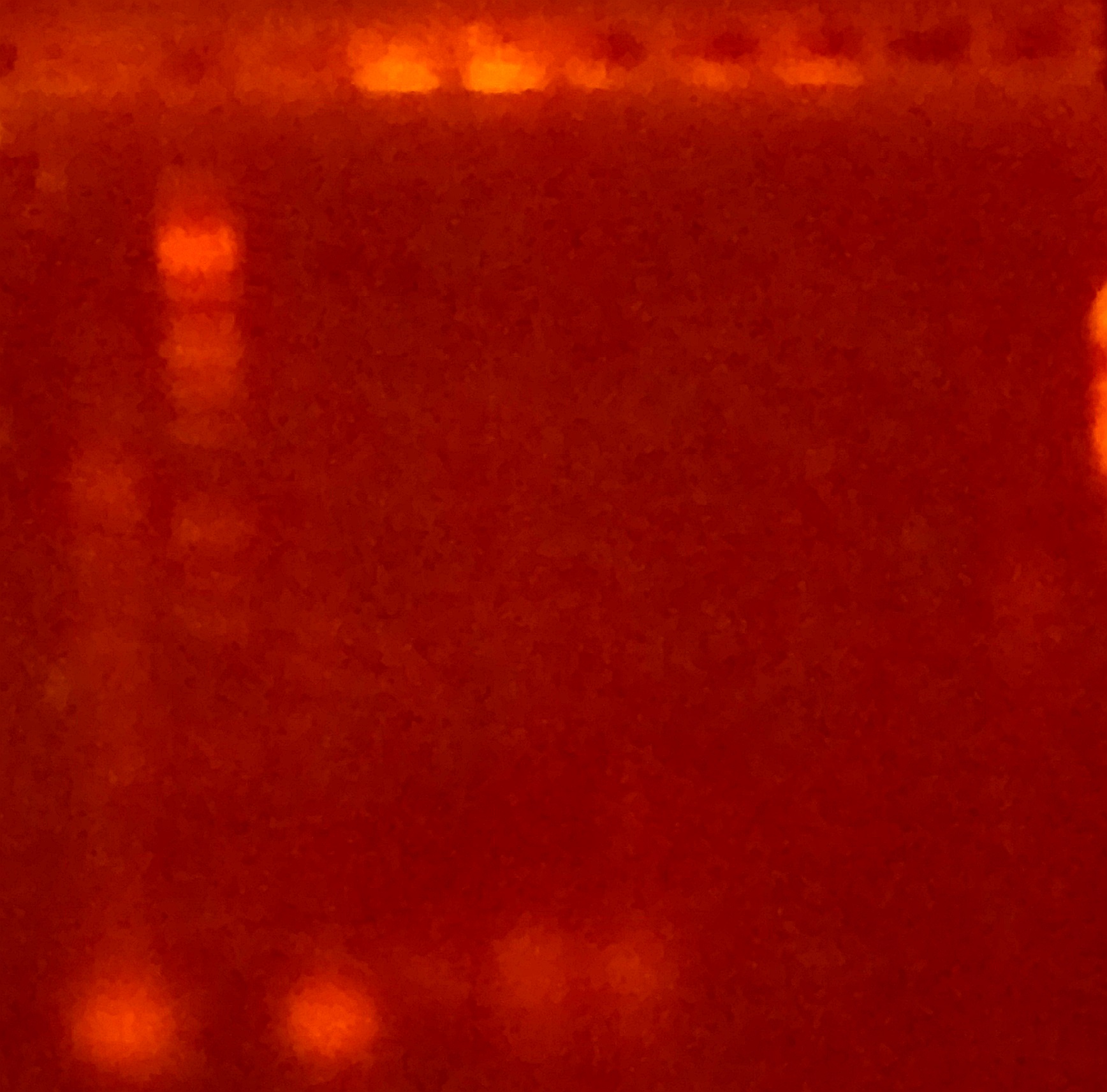
For construct K2268004, the Gibson Assembly was apparently successful, and multiple clones were obtained. The Gibson control reaction and transformation control also worked. These clones were amplified using the standard iGEM sequencing primers, VF2 and VR, which have homology to the pSB1C3 backbone.
The first lane contains a successful colony PCR, giving a band that corresponds in size to the genes of construct K2268004 (compare with similar band from previous section). The second band is our molecular weight marker, Lambda DNA digested with EcoRI and PstI (prepared in-house).
Sequencing Verification
Text here about the PCR reactions to create the fragments for Gibson Assembly
Full sequencing results for construct K2268004
VF2 Primer result, reverse complement
GGGNACGTTACGTTGCGCATTTAACTATTAGATGCGTCGAATCACGAGGCAGGAATTTCAGATAAAAAAAATCCTTAGCT TTCGCTAAGGATGATTTCTGGAATTCGCGGCCGCTTCTAGAGTAACACCGTGCGTGTTGACTATTTTACCTCTGGCGGTG ATAATGGTTGCGCTAGCAAAGAGGAGAAAGCTTATGACCATGATTACGGATTCACTGGCCGTCGTTTTACAACTCGTGAC TGGGAAAACCCTGGCGTTACCCAACTTAATCGCCTTGCAGCACATCCCCCTTTCGCCAGCTGGCGTAATAGCGAAGAGGC CCGCACCGATCGCCCTTCCCAACAGTTGCGCAGCCTGAATGGCGAAT
3.2_LinkR Primer result, reverse complement
ATCACCAGCTCCCGTCTTTCATTGCATACGAAATTCCGGAGAGCATTCATCAGGCGGGCA AGAAATGTGAATAAAGGCCGAATAAAACTTGTGCTTATTTTTCTTTACGGTCTTTAAAAA GGCCGTAATATCCAACTGAACGGTCTGGTTATAGGTACATTGAGCACTGACTGAAATGCC CTCAAAATGTTCTTTACGATGCCATTGGGATATATCACCGGTGGTATATCCAGTGATTTT TTTCTCCATTTTAGCTTCCTTAGCTCCTGAAAATCTCGATAACTCAAAAAATACGCCCGG TAGTGATCTTATTTCATTATGGTGAAAGTTGGAACCTCTTACGTGCCCGATCAACTCGAG TGCCACCTGACGTCTAAGAACCCATTATTATCGTGACATTAACCTATAAAAATAGGCGTA TCACGAGGCAGAATTTCAGATAAAAAAAATCCTTAGCTTTCGCTAAGGATGATTTCTGGA ATTCGCGGCCGCTTCTAGAGTAACACCGTGCGTGTTGACTATTTTACCTCTGGCGGTGAT AATGGTTGCGCTAGCAAAGAGGAGAAAGCTTATGACCATGATTACGGATTCACTGGCCGT CGTTTTACAACGTCGTGACTGGGAAAACCCTGGCGTTACCCAACTTAATCGCCTTGCAGC ACATCCCCCTTTCGCCAGCTGGCGTAATAGCGAAGAGGCCCGCACCGATCGCCCTTCCCA ACAGAGGGCAGCTTGAATGGCGAATGGGCTTTGCCTGGTTTTTCGAATTCTTCCGCAACT TACTGCGTAGCGNA
3.2_LinkF Primer result
NTCCATGATAAGTACTCGCACTTTCTCAGCAGTCATCAGTTCGCCGCGCCAAACGTCTCTTCAGGCCACTGACTAGCGAT AACTTTCCCCACAACGGAACAACTCTCATTGCATGGGATCATTGGGTACTGTGGGTTTAGTGGTTGTAAAAACACCTGAC CGCTATCCCTGATCAGTTTCTTGAAGGTAAACTCATCACCCCCAAGTCTGGCTATGCAGAAATCACCTGGCTCAACAGCC TGCTCAGGGTCAACGAGAATTAACATTCCGTCAGGAAAGCTTGGCTTGGAGCCTGTTGGTGCGGTCATGGAATTACCTTC AACCTCAAGCCAGAATGCAGAATCACTGGCTTTTTTGGTTGTGCTTACCCATCTCTCCGCATCACCTTTGGTAAAGGTTC TAAGCTAAGGTGAGAACATCCCTGCCTGAACATGAGAAAAAACAGGGTACTCATACTCACTTCTAAGTGACGGCTGCATA CTAACCGCTTCATACATCTCGTAGATTTCTCTGGCGATTGAAGGGCTAAATTCTTCAACGCTAACTTTGAGAATTTTTGC AAGCAATGCGGCGTTATAAGCATTTAATGCATTGATGCCATTAAATAAAGCACCAACGCCTGACTGCCCCATCCCCATCT TGTCTGCGACAGATTCCTGGGATAAGCCAAGTTCATTTTTCTTTTTTTCATAAATTGCTTTAAGGCGACGTGCGTCCTCA AGCTGCTCTTGTGTTAATGGTTTCTTTTTTGTGCTCATAAGCTTTCTTCCTCTTTGCTAGCTGTGCTCAGTATCTTGTTA TCCGCTCACAATTTACTAGTAGCGGCCGCTGCAGGCCCGGCAAAAAAGGCAAGGGGTCACCACCCTGCCCTTTTTCTTTA AAACCGAAAAGATTACTTCGCGTTATGCAGGGTTCCTCGCTCACTGACTCGCTGCGCTCGGGCGTCGGCTGCGGCGGAGC GGATCAGCTCACTCAAGCGGTAATACGGTATCACAGAATCAGGGATACCGCAGAAGAACATGTGAGCAAAGGCAGCAAAG GCAGGACTCGTAAAAGCCGCGTGCTGGCGATTCACAAGGCTCGCCCCCTGACGGCACTACAAAATCGACGCTCTAGGCAG AGGTGCGAACCGCACGGACATAAAGGATCCAGC
* VR Primer result, reverse complement
ATGATTCGGATCCCTGGCCGTGTTTACACGTTCGGAACTGGGAAACCCTGCGTACCAACT AATCCCTTGCAGCACATCCCTTTCGCAGCTGCGTAATAGCGAAGAGGCCGCCCGATCGCC CTTCCCAACAGTTGCGCAGCCTGAATGGCGAATGCGCTTGCCTGGTTTCCGGCACCAGAA GCGGTGCCGAAAGCTGGCTGGAGTAATATTATTACGCCACCAGCGCATAGTTTTCATCGT TCGCGCGCAAACGTCTCTTCAGGCACTGACTAGCGATAACTTTCCCCACAACGGAACACC TCTCATTGCATGGGATCATTGGGTACTGTGGGTTTAGTGGTTGTAAAAACACCTGACCGC TATCCCTGATCAGTTTCTTGAAGGTAAACTCATCACCCCCAAGTCTGGCTATGCAGAAAT CACCTGGCTCAACAGCCTGCTCAGGGTCAACGAGAATTAACATTCCGTCAGGAAAGCTTG GCTTGGAGCCTGTTGGTGCGGTCATGGAATTACCTTCAACCTCAAGCCAGAATGCAGAAT CACTGGCTTTTTTGGTTGTGCTTACCCATCTCTCCGCATCACCTTTGGTAAAGGTTCTAA GCTAAGGTGAGAACATCCCTGCCTGAACATGAGAAAAAACAGGGTACTCATACTCACTTC TAAGTGACGGCTGCATACTAACCGCTTCATACATCTCGTAGATTTCTCTGGCGATTGAAG GGCTAAATTCTTCAACGCTAACTTTGAGAATTTTTGCAAGCAATGCGGCGTTATAAGCAT TTAATGCATTGATGCCATTAAATAAAGCACCAACGCCTGACTGCCCCATCCCCATCTTGT CTGCGACAGATTCCTGGGATAAGCCAAGTTCATTTTTCTTTTTTTCATAAATTGCTTTAA GGCGACGTGCGTCCTCAAGCTGCTCTTGTGTTAATGGTTTCTTTTTTGTGCTCATAAGCG TTAGTATAACATTGAAGAGCTATTCGCATCACACGGATCTCCGAGCGCATTGAGCCATTG AACGAGGAATCGGAAGAGCGCCCATTAATCAATCGCCCCTCGCGCGGGGAGAGGCGGTTT GCGTATTGGGCGCTCTGCCCCTCACTCGCTCCCTCTGGAAACTGCGGCAGTACGTTGCGC GGAATGTAAGG
The sequencing primers that were used included the 3.2 Link F Primer (ggtgccggaaagctggctggagtaataaTtattacgccaccagcgcatag) and 3.2 Link R Primer (ctatgcgctggtggcgtaataAttattactccagccagctttccggcacc)